Laminin
Laminin (British: laminin) is huge protein constituting the basement membrane of the extracellular matrix. I promote the multicell system, organization construction and the maintenance, cell adhesion, cell movement, cell proliferation, and a cancer cell and relations are close. It develops for blastogenic initial (2 cells period) [1].
Table of contents
Discovery
The element of the basement membrane understood that type IV collagen, proteoglycan, laminin were three major elements now, but did not readily know it when I saw it historically. Because, by morphologic technique (an optical microscope and electron microscope), the existence of the basement membrane was confirmed, but there was little content organizing, and it was difficult to refine a shape to an index for a film without an index of the enzyme activity. Therefore I isolated quantity as I could analyze it and was not able to be refined biochemically.
In 1977, George Martin (G.R. Martin) of the cranial face research institute and others got the beginning national dentistry of United States, NIH. I found EHS sarcoma (I read with E H S.Engelbreth-Holm-Swarm sarcoma) that a mouse was implantable subcutaneously being the rare sarcoma which composed a basement membrane ingredient in large quantities [2]. EHS sarcoma of 5-15g is provided with wet weight per one mouse. It was to understand it later, but, for the laminin refinement, the cryopreservation was made, too.
In 1979, Rupert ティンプル (Rupert Timpl [3]) of the Max Karl Ernst Ludwig Planck biochemistry research institute of Germany purified new protein from this EHS sarcoma in cooperation with Martin and, for "lamina of one layer, ベーサルラミナ (basal lamina, basal lamina) of the basement membrane," named it laminin (laminin) [4].
In addition, as for the name basement membrane (basement membrane), some definitions are confused in the English zone. So even Wikipedia Japanese edition is confused. Here of "basement membrane" of the Wikipedia English version adopt "the basement membrane consists of two levels of ベーサル lamina (basal lamina) and the レティキュラ lamina" (線維細網板). The ingredient of the レティキュラ lamina is type I and type V collagen mainly.
The ベーサル lamina (basal lamina) is made of two levels of a transparent board (lamina lucida, lamina ルシダ) observed with an electron microscope more and the minute board (lamina densa, lamina デンサ) and is estimated when the laminin exists in the domain of the transparence board. In addition, it is estimated that type IV collagen and the proteoglycan exist in the domain of the minute board.
Structure
Heterotrimeric structure
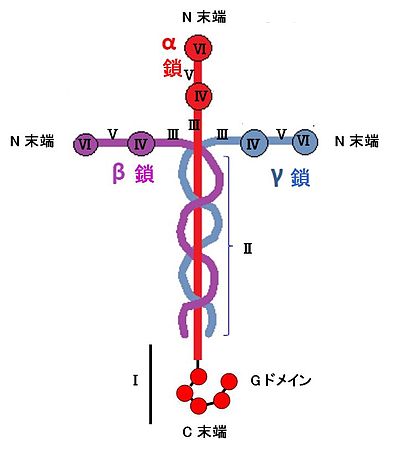
The laminin takes heterotrimeric structure to have alpha chain, β chain, γ chain by one of them each to show it in figure 1. Laminin - 111 (laminin derived from mouse EHS sarcoma) is 1:1 by (disulfide combination) S-S-binding the polypeptide chain of three of α 1 chain of molecular weight 440kDa, beta 1 chain of 220kDa, γ 1 chain of 205kDa: It is the huge glycoprotein which associated to 1. I include approximately 15% of sugar, approximately 30% of alpha helix, approximately 15% of beta sheet.
Cross-shaped model
In 1981, Swiss you gene Engel (Jürgen Engel) observed single laminin molecules with an electron microscope for the first time. I proposed "a cross-shaped model of the laminin" in one (one long arm 77nm, three short arms 37nm) of that the asymmetric "cross" showed the form [5].
CDNA base sequence
In 1987, Yoshihiko Yamada (reed Yamada) of national dentistry, skull face research institute of United States, NIH and others elucidated the cDNA base sequence of laminin β chain and γ chain using a recombinant DNA technology, and was decided in primary structure of the protein [6]; [7]. I elucidated the cDNA base sequence of the alpha chain in 1988, the following day and decided all primary structure of current laminin -111 [8].
Cell adhesion activity
By technique identical to what was studied in fibronectin in 1980, Martin found cell adhesion activity to laminin [9]. Interestingly, the laminin glued an epithelium cell and nerve cells together whereas fibronectin glued a fibroblast together. As a receptor on the cell side was analyzed as for this later, it was, and the exception mentioned above was found in the problem that which cell developed in with which receptor a lot, and, as for "the laminin, making it it did not do a meaning the type called an epithelium cell and nerve cells with a fibroblast fibronectin".
However, the laminin was conscious of for the second cell adhesion numerator next to the fibronectin, and a way of thinking and experiment technique that were effective for a study of the fibronectin were used many. It is examination of the cell adhesion activity of the laminin piece with the proteolytic enzyme, cDNA sequencing and cell adhesion motif analysis, the active official approval with the organic synthetic peptide. Thanks to grace, the cell adhesion active site of the laminin was elucidated rapidly.
Surprisingly, several places of cell adhesion parts were to the laminin. I show some cell adhesion sequence by amino acid one character notation as follows. In addition, there is a difficult point that cell adhesion activity is weak to these synthetic peptides.
- YIGSR: With the sequence that became most famous, it is in domain III of β chain. It is YIGSR equal to an amino acid number 929-933 (Tyr-Ile-Gly-Ser-Arg, tyrosine - isoleucine - glycine - serine - arginine).
- PDSGR: It is in domain III of β chain.
- RYVVLPR: It is in domain III of β chain.
- LRE: It is in domain I of β chain.
- IKVAV: It is in domain I of the alpha chain.
- RNIAEIIKDI: It is in domain I of γ chain.
- RGD: The RGD motif discovered in fibronectin was found in domain III of the laminin alpha chain. But it does not function for cell adhesion when the molecules are usually with laminin and do not break whether laminin is denatured.
In addition, the field water group justice (I quit the cause without seeing it) performs peptide composition cyclopedically and finds 22 cell adhesion activity sequence including the above in total [10].
Laminin binding molecule
As for the laminin binding molecule, "extracellular matrix molecules" forming an extracellular matrix together can classify roughly 2 to "a laminin receptor" of the cell surface.
1.Extracellular matrix molecules
I show extracellular matrix molecules and a binding site on the laminin side to be connected.
- Laminin: I meet by oneself. Domain V of β chain, domain V of γ chain
- α dystroglycan (α-dystroglycan, clay Nin cranin): G domain of domain I of the alpha chain
- Heparan sulfate (heparan sulfates), heparin
- Sulfation glucide and lipid (sulfated glycolipids): Domain VI of β chain, domain VI of γ chain
- Type IV collagen (type IV collagen)
- Pearl perception (perlecan)
- Agrin (agrin)
- ナイドジェン (nidogen): Domain III of the alpha chain
2. Laminin receptor
As for the laminin receptor, "integrin" can classify roughly 2 to "non-integrin".
It is integrin 2-1
Because the laminin was conscious of for the second cell adhesion numerator next to the fibronectin, search for receptor of the cell surface was carried out in the same way as fibronectin. A fibronectin receptor and similar integrin were discovered as a laminin receptor. As of August, 2013, 11 kinds of following integrins are laminin receptors.
α 1 beta 1, α 2 beta 1, α 2 beta 2, α 3 beta 1, α 6 beta 1, α 6 β 4, α 7 beta 1, α 9 beta 1, αvβ3, αvβ5, αvβ8
It is non-integrin 2-2
The laminin receptor which was not integrin was found [11].
- 37/67kDa laminin receptor (LPR/LR) (LamR) ... 67kDa laminin receptor (67-LR, 67LR) was discovered first [12]. I understand that protein of 37kDa consisting of 295 amino acids is a precursor afterwards when cDNA base sequence of 67-LR is elucidated. I call 37kDa 37-LRP (in P P of precursor, precursor) and am treated with 37/67 kDa laminin receptor. The active site was named peptide G in peptide, IPCNNKGAHSVGLMWWMLAR consisting of 20 amino acids of an amino acid number 161-180 turn [13].
- A laminin receptor of 120kDa was discovered in clay Nin (α dystroglycan) ... 1987 and was named cranin (clay Nin) [14]. I was proved to be molecules same as dystroglycan (dystroglycan) which seemed according to then in 1995 [15].
- Protein of 67kDa which connected アスパルタクチン (カルセクエストリン) to laminin (negative) in ... 1988 was discovered and, as a laminin receptor, was named アスパルタクチン (aspartactin). For 1,990 years, it was reported that アスパルタクチン was a molecule same as intracellular protein, カルセクエストリン (calsequestrin) of the muscular cell. When I exist in a cell, I cannot show a receptor function. And, for 1,991 years, it was said that アスパルタクチン was not a laminin receptor [16].
- Some molecules are reported in other ... others as a laminin receptor.
Domain structure
I suggest that I read the following while referring to a laminin model of figure 1. Protein usually explains on the C-terminal side from the N-terminal side, but, here, can go ahead from I of the Roman numeral to VI. In other words, I can go ahead from the C-terminal side.
Domain I (含, G domain)
LAMA1; LAMA2; LAMA3; LAMA4; LAMA5 has it when I write it with a gene in the domain of the C-terminal side of the alpha chain. It is G domain to be particularly important (globular domain). The G domain functions in cell adhesion, cell movement, signal transmission, the cytodifferentiation in the cell in the domain that is important to cell action of the laminin. It is near the C-terminus of the alpha chain and has structure repeatedly five times (LG 1, LG 2, ... LG 5). With a heparin binding site, the cell adhesion part [17]. Only LG 4 and LG 5 connect the second structure of the G domain to heparin, sulfation glucide and lipid, α dystroglycan in five G domains that are similar to pentraxin (pentraxin) [18] [17].
Domain II
Having coiled-coil of the alpha helix, there is a duty to maintain space when laminin formed basement membrane.
Domain III (= EGF-like domain)
Because there are many glycine and cysteine in domain III, there are many buckling up curves of the peptide chain. There is the repetition structure of approximately 60 amino acids having eight cysteine. Because the amino acid sequence of this repetition structure is similar to EGF and transforming growth factor alpha, is called the EGF-like domain [19]; [20] [21]. Or I am called the "EGF state (laminin-type EGF-like) of the laminin type" domain (LE). As for this "EGF state domain of the laminin type", copy numbers are various with 22 from 3 by laminin. Only the seventh "EGF state domain of the laminin type" connects laminin γ 1 of the mouse to ナイドジェン (nidogen) [22]; [20] [21].
There is not only domain III on all the laminin alpha chain, β chain, γ chain, and sequence similar to other protein is found. When write it with a gene; they are AGRIN; ATRN; ATRNL1; CELSR1; CELSR2; CELSR3; CRELD1; HSPG2; MEGF10; MEGF12; MEGF6; MEGF8; MEGF9; NSR1; NTN1; NTN2L; NTN4; NTNG1; NTNG2; RESDA1; SCARF1; SCARF2; SREC; It is STAB1; USH2A.
Domain IV (= B domain)
There is the B domain (= domain IV) to the laminin of alpha chain except α 4 of short chain Ramin and all γ chains, but there is not it to β 3 either. The function is unknown. I exist to heparan sulfate proteoglycan (HSPG2) which is other molecules, nematode laminin.
Domain V (= EGF-like domain)
It is the same as domain III.
Domain VI
Domain VI is called the LN domain in a domain in the N-terminal neighborhood. When laminin builds an extracellular matrix, the meeting with other molecules is necessary, too, but the self-association is necessary, and domain VI is a self-association domain. [23]. A disease, the muscular dystrophy that domain VI of α 2 suffered a loss hereditarily is found [24].
There is domain VI to α 3A, α 4, Ramin except γ 2. In other words, LAMA1; LAMA2; LAMA3; LAMA5; LAMB1; LAMB2; LAMB3; LAMB4; LAMC1; LAMC3 lasts. Different protein, nito phosphorus (netrins) in the extracellular matrix has this domain VI structure. In other words, it is NTN1; NTN2L; NTN4; NTNG1; NTNG2; USH2A lasts.
Discovery and naming of the laminin family
Until 1987, only laminin derived from mouse EHS sarcoma was studied as laminin.
In 1988, new protein similar to the cDNA base sequence of alpha chain of laminin molecules, β chain, γ chain a place was found in Schwann cells, a striated muscle, the basement membrane of トロホブラスト. The protein was named メローシン (merosin) [25].
In 1989, new protein similar to laminin was discovered in the synaptic cleft of the rat neuromuscular junction and took an initial of synaptic (the synapse) and was named s- laminin (s-laminin) [26]. Some laminin resemblance protein was discovered, and a name was complicated, and confusion came to show a slight it afterwards.
In 1994, main laminin researchers of the world talked, and, as a new name of the laminin, hit a number after subunit α, β, γ and classified names of the laminin integrally. For example, the laminin derived from the EHS sarcoma which I spoke is laminin - 1 so far, and the constitution subunit is α 1, beta 1, γ 1.
Three kinds of γ chains were caught in the laminin three kinds of β chains five kinds of alpha chain. Therefore, the maximum number of theoretical combinations is 5*3*3 = 45, and 45 kinds of laminin molecules are possible. However, all combinations of alpha chain, β chain, γ chains are not found. As of August, 2013, 17 kinds (19 kinds?[27]) of laminin molecules are found. In addition, 17 kinds of variety exists more than (19 kinds) because selective splicing happens on α 2 chain, α 3 chain, γ 3 chain when I write variety exactly.
However, there were two problems. Even if the constitution subunit of laminin - 1 understands it when it is α 1, beta 1, γ 1, then it is inconvenient because many researchers do not remember it when "laminin -15" is asked about と. In addition, the new laminin discovered next to laminin -15 as an essential problem becomes laminin -16 with the turn, but is short in unity because I do not cope with subunit constitution. With years, these non-unities increase.
In 2005, the glossology of a more improved model was suggested in it. Because laminin - 1 is α 1, beta 1, γ 1, let's assume it "laminin -111". Because laminin -15 is α 5, beta 2, γ 3, let's assume it "laminin -523". There are a subunit and correspondence if I name it it in this way. In addition, I can keep unity if I do it with laminin -123 without α 1, beta 2, laminin of γ 3 being found, and doing it in laminin -16 even if I do not change a turn. Laminin -16, the laminin (laminin -212/222 and laminin -522) considered that I was named laminin -17 have been already caught in an old name two [28].
List of laminin families
17 kinds (19 kinds?[27]) of pairs are caught in the laminin with three kinds γ chain three kinds of β chains five kinds of alpha chain. A function, the distribution of the laminin vary according to the kind.
In Wikipedia English version, laminin of the laminin, an item is made by each of five kinds of alpha chain, four kinds of β chains, three kinds of γ chains (as of August, 2013, β 4 identifies only a gene). Because the Wikipedia Japanese edition supported it, as for the detailed exposition, see each following items.
- Five kinds of alpha chain: LAMA1、 LAMA2、 LAMA3、 LAMA4、 LAMA5
- Four kinds of β chains: LAMB1、 LAMB2、 LAMB3、 LAMB4
- Three kinds of γ chains: LAMC1、 LAMC2、 LAMC3
Here, I show a list of whole laminin family on the following faces. There does not need to be hyphen ・" - which I added to the existing name. For example, "laminin 111" is enough for "laminin -111" [29]; [30].
| Old name | Old former name designation | Subknit | The existing name | Action |
|---|---|---|---|---|
| Laminin 1 | EHS laminin | α 1 beta 1 γ 1 | Laminin -111 | Neuritic promotion, cell movement, milk synthetic promotion of the breast cell, acetylcholine receptor meeting, nerve line binding site form |
| Laminin 2 | メローシン | α 2 beta 1 γ 1 | Laminin -211 | Neuritic promotion, the nerve line binding site formation |
| Laminin 3 | s- laminin | α 1 beta 2 γ 1 | Laminin -121 | Synapse of the neuromuscular junction |
| Laminin 4 | s- メローシン | α 2 beta 2 γ 1 | Laminin -221 | Line original junction, トロホブラスト |
| Laminin 5 | Cali Nin, エピリグリン, others | α 3 β 3 γ 2 | Laminin -332 | Epidermis blister symptom, epithelium cell adhesion, cell movement promotion, the ヘミデスモソーム formation, skin reproduction, the gap junction formation |
| Laminin 6A | k- laminin | α 3A beta 1 γ 1 | Laminin -3A11 | Epithelium |
| Laminin 7A | ks- laminin | α 3A beta 2 γ 1 | Laminin -3A21 | |
| Laminin 8 | α 4 beta 1 γ 1 | Laminin -411 | Choroidal capillary basement membrane | |
| Laminin 9 | α 4 beta 2 γ 1 | Laminin -421 | ||
| Laminin 10 | α 5 beta 1 γ 1 | Laminin -511 | Choroid plexus epithelium basement membrane | |
| Laminin 11 | α 5 beta 2 γ 1 | Laminin -521 | Schwann cells | |
| Laminin 12 | α 2 beta 1 γ 3 | Laminin -213 | ||
| Laminin 14 | α 4 beta 2 γ 3 | Laminin -423 | ||
| Laminin 15 | α 5 beta 2 γ 3 | Laminin -523 | ||
| α2β1γ2/α2β2γ2 | Laminin -212/222 | |||
| α 5 beta 2 γ 2 | Laminin -522 |
Gene knockout
I made the mouse which let a specific laminin gene make a loss (knockout) artificially and showed the symptom of the mouse on the following faces. When do not result in death even if knock out a laminin gene to show it on the face, other laminin and adhesion molecules may substitute it not what do not function in vivo [31]; [32].
The cable address of the "verge of death" column of the table "E show the death, V: survival before and after P: birth on the death and a death day for the outbreak middle".
| The verge of death | Symptom | |
|---|---|---|
| α 1 | E7 | Basement membrane loss |
| α 2 | V | Muscular dystrophy |
| α 3 | P | Foam of the skin |
| α 4 | V | Embryo bleeding, newborn baby bleeding, motor deficit |
| α 5 | E16.5 | Syndactylism symptom of outside brain fever, hand or the foot, placenta abnormality |
| Beta 1 | E5.5 | Basement membrane hypoplasia |
| Beta 2 | It is death in V, three weeks after birth | Congenital nephrotic syndrome, Pearson's syndrome |
| β 3 | V | Junctional type epidermis blister symptom (JEB) |
| β 4 | Is it unreported? | |
| γ 1 | P | Endoblast hypoplasia, basement membrane hypoplasia, motor deficit, Schwann cytodifferentiation imperfection |
| γ 2 | It is death within five days after P, life | ヘミデスモソーム development imperfection, foam of the skin |
| γ 3 | V | Normal life. Little abnormality. Extraretinal capillary increase |
Function, action of the laminin
| The correction of this knob is expected. (August, 2013) |
Basement membrane formation of the laminin
How will laminin form basement membrane? The structure has been gradually elucidated [33]; [34] [23].
A nerve and laminin
A cancer and laminin
YIGSR peptide prevents the metastasis of cancer. Refer to YIGSR. The laminin peptide except YIGSR acts on a cancer cell, too [35].
A disease and laminin
The disease in conjunction with the laminin is classified roughly into genetic deletion, a disease by the quantitative qualitative abnormality of laminin in itself such as the variation and an autoimmune disease to assume laminin a target antigen.
Deletion and the nonsense mutation of laminin α 2 chain cause some congenital muscular dystrophy (). They present with a junctional type epidermis blister symptom () by the deletion of laminin α 3 chain. In addition, the deletion of laminin α 4 chain results in the hypoplasia of the capillary [36]. Pearson's syndrome indicating the characteristic phenotype such as nephrotic syndrome and the small pupil symptom congenital as for the deletion of the laminin beta 2 chain (British :Pierson syndrome[37]) をきたす [38] [39].
The autoantibody which assumes laminin γ 1 chain [40], laminin -332 [41] a target antigen with a part of the pemphigoid that is an autoimmune disease is detected, and connection with the condition of a patient is considered.
A stem cell study and laminin
In the field of the regenerative medicine using the stem cell, it is assumed urgent business in future living body application that I remove an ingredient derived from animals such as the serum added in feeder cells and a nutrient medium used in the case of culture. A report that succeeded was made by culturing an embryonic stem cell on laminin -511 from two groups in 2008 [42]; [43]. Furthermore, the former group succeeds in removing an admixture ingredient of nutrient mediums in embryonic stem cell culture system on laminin -511; prospective future development [44]; [45].
Pharmaceutical products, applied product
- Anticancer agent: I prevent the metastasis of cancer with YIGSR peptide. Refer to YIGSR.
- An anticancer agent: The development [46] of the anticancer agent using 67kDa laminin receptor (67LR) emerging in the cancer cell which had high malignancy high.
- Antibacterial peptide: When many bacteria have a laminin receptor and are infected with Homo sapiens, I am infected with Homo sapiens taking advantage of laminin of the Homo sapiens and the interaction of the laminin receptor. Can design the bio material with the antibacterial action by making an imitation (ミメティックス) of laminin or the laminin [47]; [48].
| The correction of this knob is expected. (August, 2013) |
Footnote, references cited
- ^ Dziadek, M; et al. (Oct 1985). "Expression of nidogen and laminin in basement membranes during mouse embryogenesis and in teratocarcinoma cells." Dev Biol. 111 (2): 372-82. doi: 10.1016/0012-1606(85) 90,491-9.
- ^ Orkin RW, Gehron P, McGoodwin EB, Martin GR, Valentine T, Swarm R. (Jan 1977). "A murine tumor producing a matrix of basement membrane". J Exp Med 145 (1): 204-220. PMC 2180589. It is . PMID 830788
- ^ https://de.wikipedia.org/wiki/Rupert_Timpl
- ^ Timpl R, Rohde H, Robey PG, Rennard SI, Foidart JM, Martin GR (Oct 1979). "Laminin--a glycoprotein from basement membranes." J Biol Chem 254 (19): 9933-9937. PMID 114518.
- ^ Engel J, Odermatt E, Engel A, Madri JA, Furthmayr H, Rohde H, Timpl R. (July 1981). "Shapes, domain organizations and flexibility of laminin and fibronectin, two multifunctional proteins of the extracellular matrix." J. Mol. Biol. 150 (1): 97-120. PMID 6795355.
- ^ Sasaki M, Kato S, Kohno K, Martin GR, Yamada Y (Feb 1987). "Sequence of the cDNA encoding the laminin B1 chain reveals a multidomain protein containing cysteine-rich repeats". Proc Natl Acad Sci U S A 84 (4): 935-939. PMC 304334. It is . PMID 3493487
- ^ Sasaki M, Yamada Y (Dec 1987). "The laminin B2 chain has a multidomain structure homologous to the B1 chain." J Biol Chem 262 (35): 17,111-17,117. PMID 3680290.
- ^ Sasaki M, Kleinman HK, Huber H, Deutzmann R, Yamada Y (Nov 1988). "Laminin, a multidomain protein. The A chain has a unique globular domain and homology with the basement membrane proteoglycan and the laminin B chains." J Biol Chem 263 (32): 16,536-16,544. PMID 3182802.
- ^ Terranova VP, Rohrbach DH, Martin GR. (Dec 1980). "Role of laminin in the attachment of PAM 212 (epithelial) cells to basement membrane collagen." Cell 22 (3): 719-726.
- ^ field water group justice (2000). "How many active sites does the laminin have?". Protein nucleic acid enzyme 45 (15): 2475-2482.
- ^ Mecham RP (Aug 1991). "Receptors for laminin on mammalian cells." FASEB J 5 (11): 2538-2546. PMID 1651264.
- ^ Rao NC, Barsky SH, Terranova VP, Liotta LA (Mar 1983). "Isolation of a tumor cell laminin receptor." Biochem Biophys Res Commun 111 (3): 804-808. PMID 6301485.
- ^ Castronovo V, Taraboletti G, Sobel ME (Oct 1991). "Functional domains of the 67-kDa laminin receptor precursor." J Biol Chem 266 (30): 20,440-20,446. PMID 1834645.
- ^ Smalheiser NR, Schwartz NB (Sep 1987). "Cranin: a laminin-binding protein of cell membranes". Proc Natl Acad Sci U S A 84 (18): 6457-6461. PMC 299096. It is . PMID 2957695
- ^ Neil R. Smalheiser, Edward Kim (Jun 1995). "Purification of Cranin, a Laminin Binding Membrane Protein. IDENTITY WITH DYSTROGLYCAN AND REASSESSMENT OF ITS CARBOHYDRATE MOIETIES." It is doi: 15,425-15,433 J Biol Chem 270 10.1074/jbc.270.25.15425.
- ^ Choi ES, Sullivan PD, Clegg DO (Feb 1991). "Evidence against a laminin receptor role for calsequestrin." Biochem Biophys Res Commun 174 (3): 1208-1216. PMID 1825466.
- ^ a b Tisi D, Talts JF, Timpl R, Hohenester E (April 2000). "Structure of the C-terminal laminin G-like domain pair of the laminin alpha2 chain harbouring binding sites for alpha-dystroglycan and heparin". EMBO J. 19 (7): 1432-1440. doi: 10.1093/emboj/19.7.1432. PMC 310212. It is . PMID 10747011
- ^ Beckmann G, Hanke J, Bork P, Reich JG (February 1998). "Merging extracellular domains: fold prediction for laminin G-like and amino-terminal thrombospondin-like modules based on homology to pentraxins." J. Mol. Biol. 275 (5): 725-730. doi: 10.1006/jmbi.1997.1510. PMID 9480764.
- ^ Engel J (July 1989). "EGF-like domains in extracellular matrix proteins: localized signals for growth and differentiation?." FEBS Lett. 251 (1-2): 1-7. doi: 10.1016/0014-5793(89) 81,417-6. PMID 2666164.
- ^ a b Stetefeld J, Mayer U, Timpl R, Huber R (April 1996). "Crystal structure of three consecutive laminin-type epidermal growth factor-like (LE) modules of laminin gamma1 chain harboring the nidogen binding site." J. Mol. Biol. 257 (3): 644-657. doi: 10.1006/jmbi.1996.0191. PMID 8648630.
- ^ a b Baumgartner R, Czisch M, Mayer U, Poschl E, Huber R, Timpl R, Holak TA (April 1996). "Structure of the nidogen binding LE module of the laminin gamma1 chain in solution." J. Mol. Biol. 257 (3): 658-668. doi: 10.1006/jmbi.1996.0192. PMID 8648631.
- ^ Mayer U, Poschl E, Gerecke DR, Wagman DW, Burgeson RE, Timpl R (May 1995). "Low nidogen affinity of laminin-5 can be attributed to two serine residues in EGF-like motif gamma 2III4." FEBS Lett. 365 (2-3): 129-132. doi: 10.1016/0014-5793(95)00438-F. PMID 7781764.
- ^ a b Yurchenco PD, Cheng YS (August 1993). "Self-assembly and calcium-binding sites in laminin. A three-arm interaction model." J. Biol. Chem. 268 (23): 17,286-17,299. PMID 8349613.
- ^ Xu H, Wu XR, Wewer UM, Engvall E (November 1994). "Murine muscular dystrophy caused by a mutation in the laminin alpha 2 (Lama2) gene." Nat. Genet. 8 (3): 297-302. doi: 10.1038/ng1194-297. PMID 7874173.
- ^ Leivo I, Engvall E (Mar 1988). "Merosin, a protein specific for basement membranes of Schwann cells, striated muscle, and trophoblast, is expressed late in nerve and muscle development". Proc Natl Acad Sci U S A 85 (5): 1544-1548. PMC 279809. It is . PMID 3278318
- ^ Hunter DD, Shah V, Merlie JP, Sanes JR. (Mar 1989). "A laminin-like adhesive protein concentrated in the synaptic cleft of the neuromuscular junction." Nature 338 (6212): 229-234. PMID 2922051.
- ^ a b Cell adhesion molecules - Brain science dictionary
- ^ Aumailley, M; et al. (Aug 2005). "A simplified laminin nomenclature." Matrix Biol. 24 (5): 326-32. doi: 10.1016/j.matbio.2005.05.006.
- ^ http://www.lm.lncc.br/nomenclatures August 10, 2013 reading
- ^ Golbert, Daiane C F; Linhares-Lacerda Leandra, Almeida Luiz G, Correa-de-Santana Eliane, de Oliveira Alice R, Mundstein Alex S, Savino Wilson, de Vasconcelos Ana T R (Jan 2011). "Laminin database: a tool to retrieve high-throughput and curated data for studies on laminins" (Old English). Nucleic Acids Res. (England) 39 (Database issue): D320-3. doi: 10.1093/nar/gkq1055. PMC 3013663. It is . PMID 21087995
- ^ Tzu J, Marinkovich MP (2008). "Bridging structure with function: structural, regulatory, and developmental role of laminins". Int J Biochem Cell Biol 40 (2): 199-214. PMC 2192629. It is . PMID 17855154
- ^ Colognato H, Yurchenco P (2000). "Form and function: the laminin family of heterotrimers." Dev. Dyn. 218 (2): 213-234. doi: 10.1002/(SICI)1097-0177(200006)218:2<213:: AID-DVDY1>3.0.CO; 2-R. PMID 10842354.
- ^ Erhard Hohenester, Peter D. Yurchenco (January 2013). "Laminins in basement membrane assembly". Cell Adh Migr 7 (1): 56-63. doi: 10.4161/cam.21831. It is . PMC 3544787
- ^ Beck K, Hunter I, Engel J (February 1990). "Structure and function of laminin: anatomy of a multidomain glycoprotein." FASEB J. 4 (2): 148-160. PMID 2404817.
- ^ Kikkawa Y, Hozumi K, Katagiri F, Nomizu M, Kleinman HK, Koblinski JE (January 2013). "Laminin-111-derived peptides and cancer". Cell Adh Migr 7 (1): 150-56-63. doi: 10.4161/cam.22827. PMC 3544779. It is . PMID 23263633
- ^ Thyboll, J; et al. (Feb 2002). "Deletion of the laminin alpha4 chain leads to impaired microvessel maturation." Mol Cell Biol. 22 (4): 1194-202. PMID 11809810.
- ^ OMIM#609049 Pierson syndrome(2012/3/19 reading)
- ^ Zenker, M; et al. (Nov 2004). "Human laminin beta2 deficiency causes congenital nephrosis with mesangial sclerosis and distinct eye abnormalities." Hum Mol Genet. 13 (21): 2625-32. PMID 15367484.
- ^ Matejas, V; et al. (Sep 2010). "Mutations in the human laminin beta2 (LAMB2) gene and the associated phenotypic spectrum." Hum Mutat. 31 (9): 992-1002. PMID 20556798.
- ^ Dainichi, T; et al. (Feb 2009). "Anti-laminin gamma-1 pemphigoid." Proc Natl Acad Sci U S A. 106 (8): 2800-5. PMID 19196964.
- ^ Hayashi, I; et al. (2011 published online). "Mucous membrane pemphigoid with generalized blisters: IgA and IgG autoantibodies target both laminin-332 and type XVII collagen." Br J Dermatol.. doi: 10.1111/j.1365-2133.2011.10776.x. PMID 22182184.
- ^ Domogatskaya, A; et al. (Nov 2008). "Laminin-511 but not -332, -111, or -411 enables mouse embryonic stem cell self-renewal in vitro." Stem Cells. 26 (11): 2800-9.
- ^ Miyazaki, T; et al. (Oct 2008). "Recombinant human laminin isoforms can support the undifferentiated growth of human embryonic stem cells." Biochem Biophys Res Commun. 375 (1): 27-32. doi: 10.1016/j.bbrc.2008.07.111.
- ^ Rodin, S; et al. (Jun 2010). "Long-term self-renewal of human pluripotent stem cells on human recombinant laminin-511." Nat Biotechnol. 28 (6): 611-5. doi: 10.1038/nbt.1620.
- ^ Karl Tryggvason (May 2013). "Biologically Relevant Laminins. Key to Derivation and Culture of Stem Cells and Differentiated Cells Lies with These ECM Proteins". Genetic Engineering & Biotechnology News 33 (9).
- ^ patent laboratory
- ^ Kobayashi N, Yoshida T (2007). "Binding sites on laminin receptors as components for antibiotics." Protein Pept Lett 14 (1): 33-36. PMID 17266648.
- ^ Naoko Kobayashi, Tetsuhiko Yoshida (2009). "A design of the antibacterial peptide based on the laminin binding site of the laminin receptor". It is . Eastern Asia composition study annual report
Overall references
- Masao Hayashi (2001). The world of new cell adhesion molecules. Tokyo: Sheep soil company. ISBN 9784897063270.
- Colognato H, Yurchenco P (2000). "Form and function: the laminin family of heterotrimers." Dev. Dyn. 218 (2): 213-234. doi: 10.1002/(SICI)1097-0177(200006)218:2<213:: AID-DVDY1>3.0.CO; 2-R. PMID 10842354.
- Aumailley M. (Jan-Feb 2013). "The laminin family". Cell Adh Migr 7 (1): 48-55. PMC 3544786. It is . PMID 23263632
Allied item
Outside link
- laminin - the US National Library of Medicine Medical Subject Headings (MeSH)
- Variety and integrin signaling August 13, 2013 reading of the Kiyotoshi Sekiguchi extracellular matrix
- I read it on study summary August 11, 2013 of Yamato Yoshikawa
- Laminin project (English) August 9, 2013 reading
- The Laminin Protein (English) August 9, 2013 reading
- Laminin Family | Cell Migration Knowledgebase (English) August 9, 2013 reading
- How I learned to love laminin - YouTube animation (English) 53 minutes 31 seconds. Lecture "How I learned to love laminin" August 9, 2013 reading of the Hohenester professor of Imperial College London
This article is taken from the Japanese Wikipedia Laminin
This article is distributed by cc-by-sa or GFDL license in accordance with the provisions of Wikipedia.
In addition, Tranpedia is simply not responsible for any show is only by translating the writings of foreign licenses that are compatible with CC-BY-SA license information.
0 개의 댓글:
댓글 쓰기